Metallodrugs
Functionalized Platinum(IV) Anticancer Compounds
One strategy to reduce toxicities and side-effects is to improve drug delivery by using targeting methods to enhance the selectivity. At the molecular level, platinum(IV) carboxylate complexes represent suitable scaffolds, since they can be readily derived from classical platinum(II) drugs without altering the pharmacophore. Upon cell entry, they are reduced by intracellular biomolecules such as glutathione, to yield cytotoxic platinum(II) moieties while releasing the axial ligands. The general strategy to prepare these complexes is shown below.
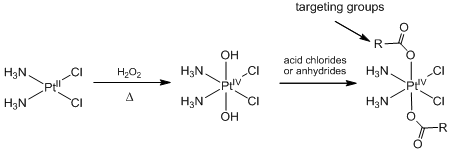
Preparation of platinum(IV) carboxylates from platinum(II) drugs
The nature of the ligand sphere affects the reduction potential of the complexes and can be tuned to achieve optimal release of the platinum(II) moiety. An orally-active platinum(IV) prodrug, satraplatin, is currently undergoing phase III clinical trials for hormone refractory prostate cancer. Other platinum(IV) compounds with functionalised ligands have also been harnessed to defeat glutathione-transferase mediated drug resistance, target estrogen receptor-positive breast cancer and for photochemotherapy.
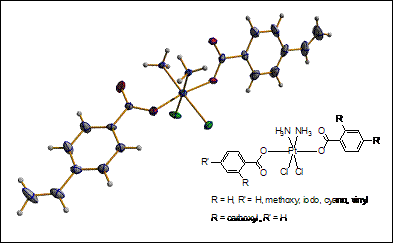
Highly hydrophobic platinum(IV) carboxylates

Loading content... Please wait a moment.
